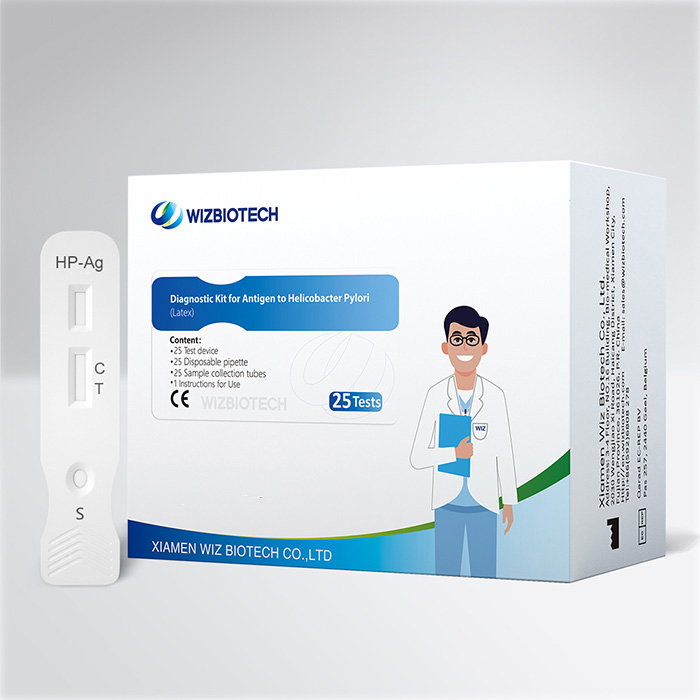
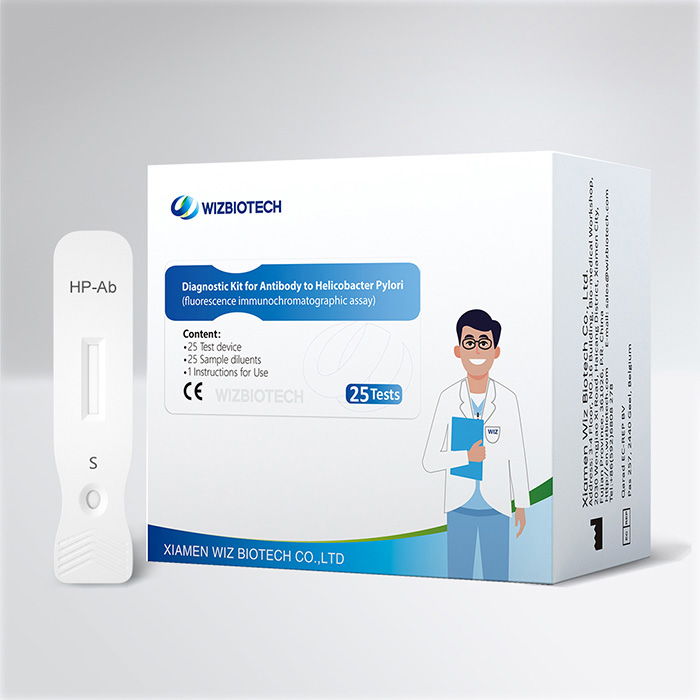
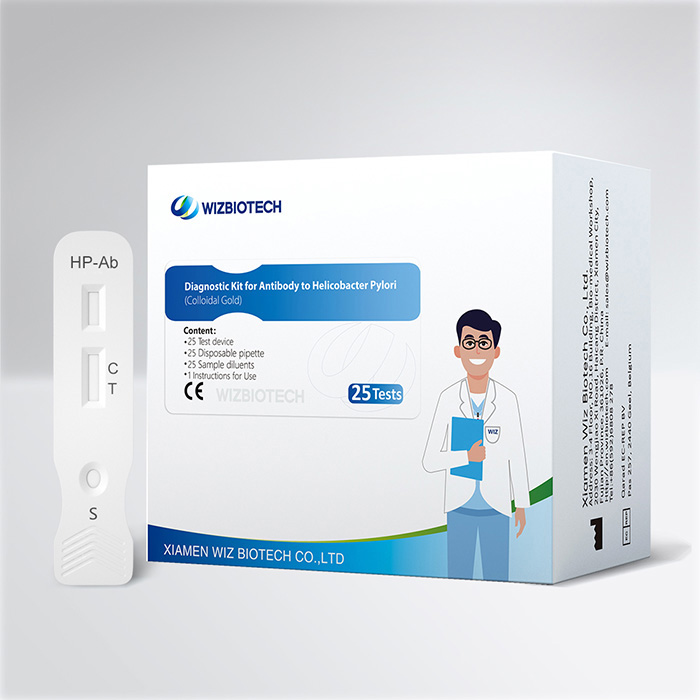
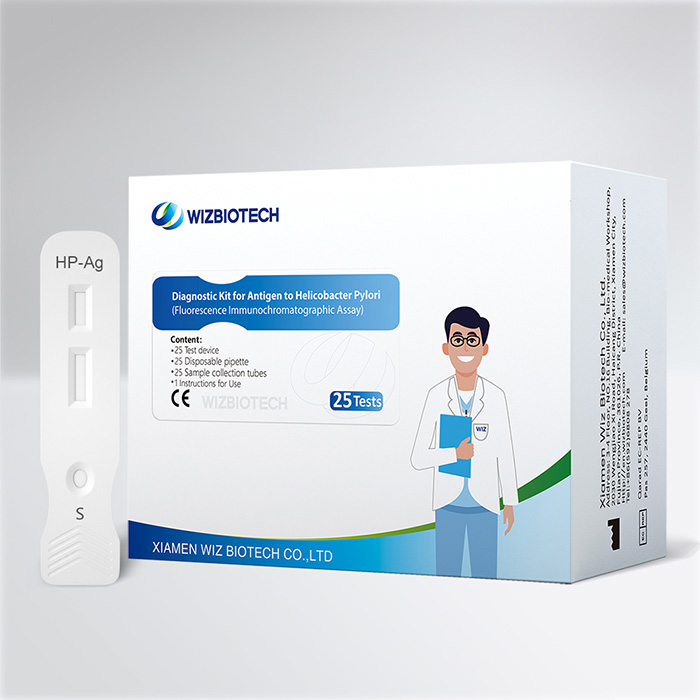
Bluttestkit zur Magenkrebsvorsorge gegen den H-Pylori-Antikörpersubtyp

- Wizbiotech
- WAS, UKCA
- China
Dieses Kit dient zum qualitativen In-vitro-Nachweis von Urease-Antikörpern, CagA-Antikörpern und VacA-Antikörpern gegen Helicobacter pylori in menschlichen Vollblut-, Serum- oder Plasmaproben und eignet sich zur zusätzlichen Diagnose einer HP-Infektion sowie zur Identifizierung des Typs der Helicobacter-pylori-Infektion eines Patienten.
Beschreibung
Helicobacter pylori kann anhand der Expression von CagA und VacA in zwei Typen unterteilt werden: Typ I ist ein toxigener Stamm (mit Expression von sowohl CagA als auch VacA oder einem von beiden), der hoch pathogen ist und leicht Magenerkrankungen verursacht; Typ II ist ein atoxigener HP (ohne Expression von sowohl CagA als auch VacA), der weniger toxisch ist und bei einer Infektion normalerweise keine klinischen Symptome aufweist.
Vorteile
Diagnostischer Wert: Der H. pylori-Antikörpertest hilft dem Gesundheitspersonal dabei, zu diagnostizieren, mit welchem Typ von HP der Patient infiziert ist. Er liefert wichtige Informationen für die klinische Entscheidungsfindung, einschließlich Behandlungs- und Managementoptionen.
Komfortabel: Der H. pylori-Antikörper-Subtyptest erfordert nur eine einzige Blutprobe und ist einfach durchzuführen, was ihn sowohl für Patienten als auch für medizinisches Personal zu einer bequemen Option macht.
Produktspezifikationen
Verfahren | Fluoreszenz-Immunchromatographischer Test |
Probentyp | Vollblut/Serum/Plasma |
| Zeit bis zum Ergebnis | 15 Minuten |
| Lagerung | 2 bis 30 °C |
| Haltbarkeit | 24 Monate ab Herstellungsdatum |
Kit Größe | 1/5/20/25 Prüfungen |
※ Weitere Produktinformationen finden Sie in der Packungsbeilage.
Produktleistung
WIZ Ergebnisse von Urease-Antikörper | Testergebnis des Referenzreagenzes | Positive Koinzidenzrate: 94,39 % (95 % KI 90,46 % – 96,76 %) Negative Koinzidenzrate: 99,32 % (95 % KI 96,28 % – 99,88 %) Gesamtübereinstimmungsrate: 96,41 % (95 % KI 93,95 % – 97,89 %) | ||
| Positiv | Negativ | Gesamt | ||
| Positiv | 202 | 1 | 203 | |
| Negativ | 12 | 147 | 159 | |
| Gesamt | 214 | 148 | 362 | |
WIZ Ergebnisse von CagAAntikörper | Testergebnis des Referenzreagenzes | Positive Koinzidenzrate: 95,28 % (95 % KI 89,43 % – 97,97 %) Negative Koinzidenzrate: 97,66 % (95 % KI 94,98 % – 98,92 %) Gesamtübereinstimmungsrate: 96,96 % (95 % KI 94,64 % – 98,29 %) | ||
| Positiv | Negativ | Gesamt | ||
| Positiv | 101 | 6 | 107 | |
| Negativ | 5 | 250 | 255 | |
| Gesamt | 106 | 256 | 362 | |
WIZ Ergebnisse von KuhAntikörper | Testergebnis des Referenzreagenzes | Positive Koinzidenzrate: 94,23 % (95 % KI 87,98 % – 97,33 %) Negative Koinzidenzrate: 97,29 % (95 % KI 94,51 % – 98,68 %) Gesamtübereinstimmungsrate:96,41 % (95 % KI 93,95 % – 97,89 %) | ||
| Positiv | Negativ | Gesamt | ||
| Positiv | 98 | 7 | 105 | |
| Negativ | 6 | 251 | 257 | |
| Gesamt | 104 | 258 | 362 | |
Anwendungen
Ambulantes Notfalllabor
Klinische Abteilungen
Gemeindekrankenhaus
Laborabteilungen
Gesundheitsmanagementzentrum
Klinik
Zertifizierungen